Table of Contents
ToggleAn organic acid with a carboxyl group (C(=O)OH) linked to an R-group is known as a carboxylic acid. A carboxylic acid has the general formula RCOOH or RCO2H, where R refers to the alkyl, alkenyl, aryl, or other group. Amino acids and fatty acids are two important examples of carboxylic acid.
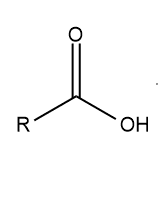
Test for carboxylic acid
Carboxylic acids are primarily identified by spectroscopy and solubility tests. However, a few classification tests can be used to confirm the presence of the carboxylic group.
Litmus Paper test
Place 0.2g of the unknown substance in a minimum volume of water. With the help of glass rod apply solution on the blue litmus paper. A change to blue litmus into red indicates a substance to be acidic.
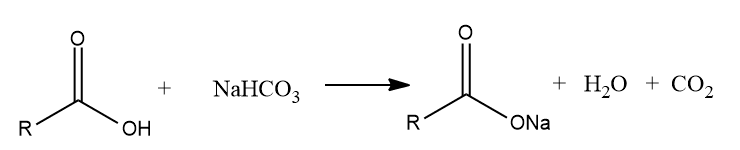
Sodium bicarbonate test
Add a few drops of liquid substance or a pinch of solid substance to 5ml of cold saturated sodium bicarbonate solution. Vigorous evolution of carbon dioxide with brisk effervescence indicates the presence of carboxylic group in the compound.
Dye acriflavine test
The dye acriflavine is used for the selective detection of the carboxylic acid group in organic compounds. Add a few drops of aqueous solution of substance to a mixture of about 3 mL % acriflavine solution and 3 mL % potassium chromate solution. The production of a violet color and a yellow to yellowish-brown precipitate when a carboxylic acid is treated with acriflavine/sodium nitrite and acriflavine/potassium chromate.
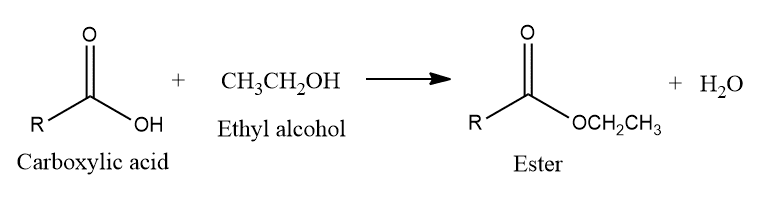
Ester test
Mix a small amount of compound with few drops of absolute ethyl alcohol and add 2 ml of conc. sulphuric acid. Heat for about 10 minutes when a fruity smell of ester indicates the presence of carboxylic acid.
After determining the carboxylic acid class, such as unsaturated acids, hydroxy acids, and so on, determine the boiling point or melting point and whether the compound is aromatic or aliphatic.









