Table of Contents
ToggleMolar conductance is another term used to express the conductivities of the electrolytic solution. it is the conductance of an electrolyte measured after dissolving one gram mole in V cc of water. Its definition, unit, formula, and its variation with dilution or concentration have been discussed below:
Define molar conductance
Molar conductance is defined as the conductance of all ions produced by dissociation of 1 gram mole of an electrolyte dissolved in V cc of the solution when electrodes are 1 cm apart and the area of the electrode is so large that the whole solution is contained between them.
It is denoted by µ and its value can be obtained by multiplying the specific conductance(κ) and volume(V in cc) containing one mole of the electrolytes as shown below:
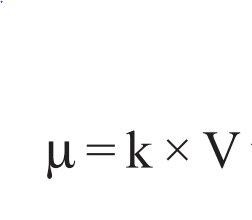
If M be the concentration in gram moles per liter then, a volume containing 1 gram moles of electrolyte becomes (1000/M). Therefore, using the value of V= 1000/M, the above equation becomes,
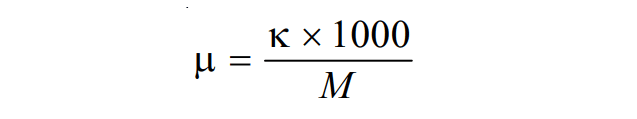
This is the relation between molar conductance and specific conductance.
Molar conductance at infinite dilution
At infinite dilution, all the electrolyte including strong as well as weak electrolytes tends to undergo complete ionization. On the other hand, at infinite dilution, the ions are far from each other and the interionic interaction is very low. Therefore, the value of molar conductivity is maximum at infinite dilution. The value of molar conductivity becomes maximum at infinite dilution, such value is called limiting molar conductance.
Unit of molar conductance
The unit of molar conductance can be derived as:
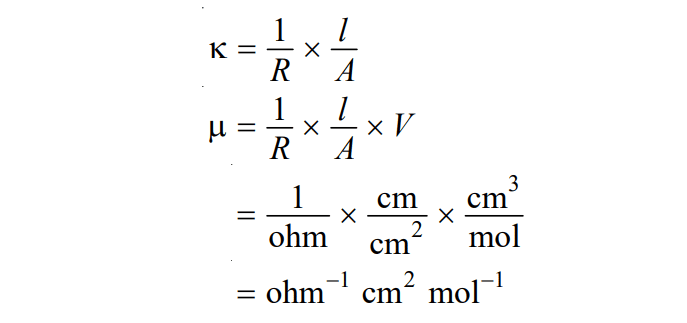
Therefore, the unit of molar conductance is ohm-1 cm2 mol-1 or mho cm2 mol-1 or S cm2 mol-1. The SI unit of molar conductance is S m2 mol-1
Molar conductance formula
The formula for molar conductance is given below:
µ= κ × (1000/M)
Relation between molar conductance and equivalent conductance
We know, the expression of equivalent conductance is given as
Λ= κ × (1000/N) …………………………………………..(a)
And, the expression of molar conductance is given as
µ= κ × (1000/M) ……………………………………………..(b)
From equations (a) and (b), we get,
Λ /µ =M/N
Λ= µ × (M/N)
This is the relation between molar conductance and equivalent conductance.
Why molar conductance increases with dilution
The molar conductance depends on the number of ions and ionic mobility. On dilution, the degree of ionization increases, and the interionic force decrease hence ionic mobility increases. Therefore, molar conductance increases dilution.
Difference between specific conductance and molar conductance
Specific conductance is the conductance offered by a one cm cube of a solution. But, Molar conductance is the conductance of an electrolyte measured after dissolving one gram mole in V cc of water.
| Specific conductance | Molar conductance |
| Specific conductance of an electrolytic solution is defined as the conductance of a solution placed between two parallel electrodes which are 1 cm distance apart and have a 1 square cm area of cross-section. | Molar conductance is defined as the conductance of all ions produced by dissociation of 1 gram mole of an electrolyte dissolved in V cc of the solution when electrodes are 1 cm apart and the area of the electrode is so large that the whole solution is contained between them. |
| It is denoted by κ. | It is denoted by µ. |
| It has a unit ohm-1 cm-1. | It has a unit ohm-1 cm2 mol-1. |
| It decreases with increasing dilution. | It increases with increasing dilution. |






