Table of Contents
ToggleBaldwin’s rules proposed by J. E. Baldwin are the kinetic rules applicable for the relative rate of ring closure reaction in alicyclic compounds. It explores the geometry of the transition states involved in the reaction. The phrases like favored and disfavored implied continuously in this rule are used in a relative sense and are not meant to express the absolute probability of a reaction occurring or not occurring.
The rules classify ring closures in three ways:
- how many atoms are in the newly formed ring
- 3 membered rings = 3
- 4 membered rings = 4
- 5 membered rings = 5, and so on.
- based on whether the bond broken during the ring closure is inside (endo) or outside (exo)
- depending on whether the electrophilic carbon is tetrahedral, trigonal, or diagonal
- Tetrahedral (Tet) for sp3 hybridized center
- Trigonal (Trig) for sp2 center
- Diagonal (Dig) for sp center
Only particular combinations of ring size and the exo/endo/dig/trig/tet factors favor the generation/formation of bonds, according to Baldwin.
Baldwin Rules
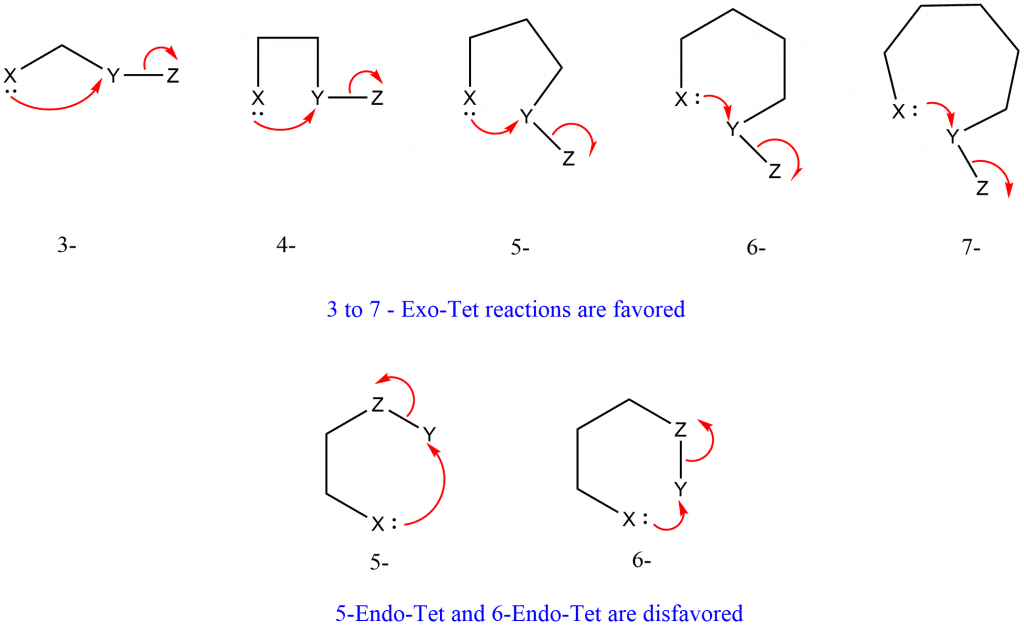
The following are Baldwin’s rules for closing 3-7 membered rings.
Rule 1: Tetrahedral System
- 3 to 7 – Exo – Tet are favored
- 5 to 6 – Endo – Tet are disfavored
Rule 2: Trigonal System
- 3 to 7 – Exo – Trig are favored
- 3 to 5 – Endo – Trig are disfavored
- 6 to 7 – Endo – Trig are favored

Rule 3: Diagonal System
- 3 to 4 – Exo – Dig are disfavored
- 5 to 7 – Exo – Dig are favored
- 3 to 7 – Endo – Dig are favored

Note: Disfavored does not imply that it is impossible to produce; rather, it indicates that it is more difficult than the favored cases.
Baldwin’s Rules example
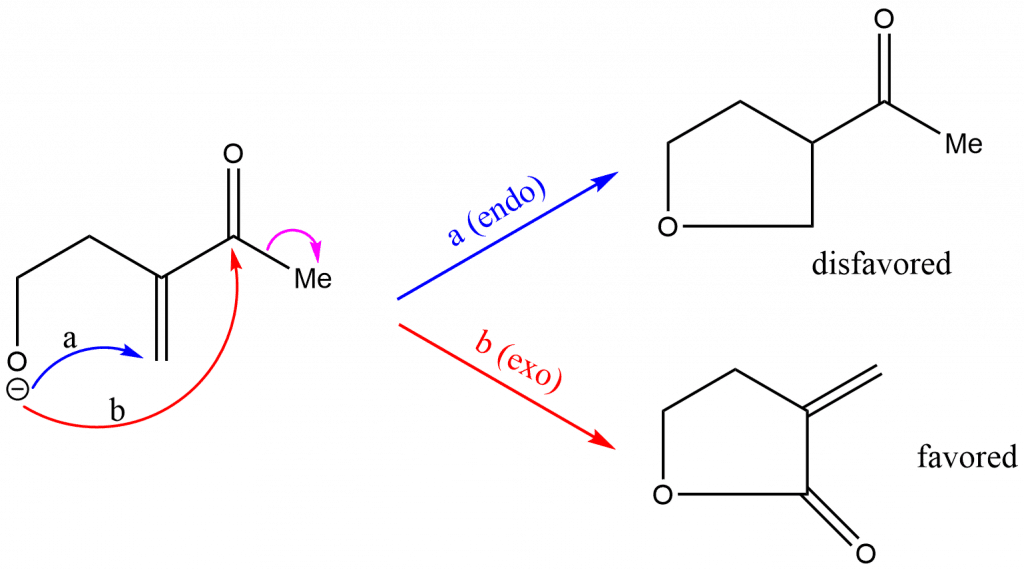
Application of Baldwin’s rule
- Applicable for aldol cyclizations via utilization of enolates.
- For clasien rearrangement reaction, and so on.
Baldwin’s Rules Video
References
- Morrison, R. T., & Boyd, R. N., Organic chemistry, Allyn and Bacon, Inc. 1987.
- March, J., Advanced Organic Chemistry, Wiley Eastern Limited, 1986.
- Baldwin, J. E., Rules for Ring Closure, J. Chem. Soc., Chem. Commun. 1976, 734. doi:10.1039/C39760000734
- Baldwin, J. E., et al, Rules for Ring Closure: Ring Formation by Conjugate Addition of Oxygen Nucleophiles, J. Org. Chem., 42(24), 3846 (1977). doi:10.1021/jo00444a011






