Table of Contents
Togglenoun, plural: glycoconjugates
Glycoconjugates are composed of carbohydrates that have been covalently joined to a non-sugar molecule, like a lipid or protein. They are frequently located on the outside of cell membranes and, for instance, take part in particular cell-to-cell recognition processes.

Glycoconjugate definition
Glycoconjugates are a group of carbohydrates, or glycans, that are covalently joined to other chemical species like proteins, peptides, lipids, and other substances. Glycosylation is the process by which glycoconjugates are formed. Glycoconjugates are biopolymers that include one or more carbohydrate units and are roughly divided into proteoglycans, glycoproteins, and glycolipids based on their structural similarities.
In simple terms, we can also explain the term glycoconjugates as Glyco means glycans (carbohydrates) and conjugates means ( linking). They participate in interactions between cells, including cell-cell recognition, interactions between cells and the matrix, and detoxification processes.
Glycoconjugate in Biochemistry
The hybrid biochemicals known as glycoconjugates comprise a carbohydrate that is chemically linked to another component, such as a peptide, protein, lipid, or molecule. Glycoconjugates, such as glycoproteins, glycolipids, and proteoglycans that carry carbohydrate antigens, are typically found on the plasma membrane of cells and are effective biomarkers at different phases of cellular differentiation.
How are glycoconjugates formed?
Glycoconjugates are formed by glycosylation and are involved in various biological processes in organisms. A carbohydrate is covalently joined to a target macromolecule—usually proteins and lipids—through the process of glycosylation. The varied purposes of this alteration are numerous. For example, without being glycosylated, some proteins do not fold appropriately.
The process of glycosylation involves the covalent attachment of a carbohydrate to a target macromolecule, usually a protein or lipid. This modification has several purposes. For instance, some proteins require glycosylation in order to fold appropriately. In other instances, oligosaccharides connected at the amide nitrogen of specific asparagine residues are required for proteins to be stable.
Examples of glycoconjugates
Proteoglycans, glycoproteins, and glycolipids are the three different types of glycoconjugates that are formed when carbohydrates are covalently joined to proteins and lipids.
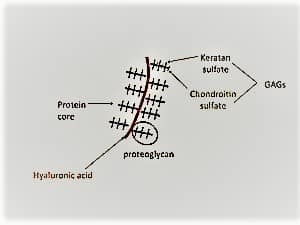
1. Proteoglycans
CHOs can modify some proteins to the point where they have more CHOs than amino acids. The collective term for proteins attached to glycosaminoglycans is proteoglycan (PGs). One or more glycosaminoglycans are connected to a core protein that makes up the PGs.
Proteoglycans are highly glycosylated proteins. A “core protein” with one or more covalently bonded glycosaminoglycan chains makes up the fundamental proteoglycan unit. The glycosaminoglycan is connected to the serine residue at the place of attachment via a tetrasaccharide bridge.
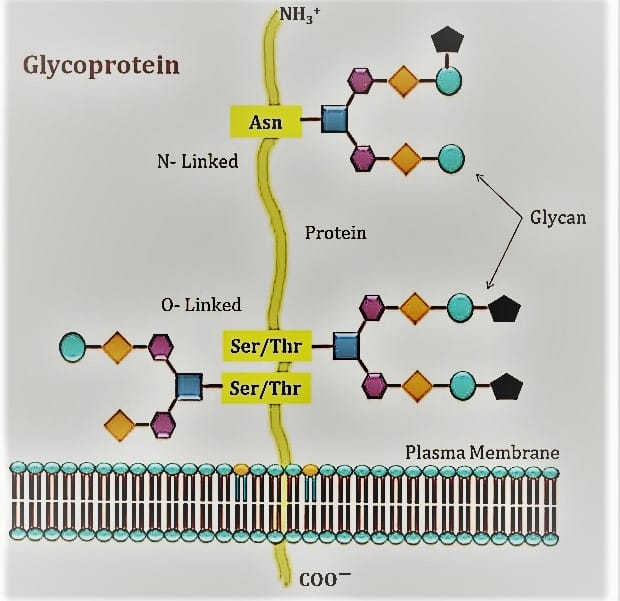
2. Glycoproteins
Glycoproteins are proteins that have oligosaccharide chains bonded to the side chains of amino acids in a covalent manner. The cotranslational or posttranslational modification involves the attachment of the carbohydrate to the protein. Glycosylation is the term for this action. Many extracellular proteins that are secreted have glycosylation. Some examples of glycoprotein are:
- collagen.
- mucins.
- transferrin.
- ceruloplasmin.
- immunoglobulins.
- antibodies.
- histocompatibility antigens.
- hormones
Glycoproteins assist in the autoimmune response. Depending on the type of carbohydrates it contains, an antibody (or glycoprotein) can attach to a certain antigen. B and T cell surface glycoproteins also bind to antigens. Our skin’s health is also maintained by glycoproteins.
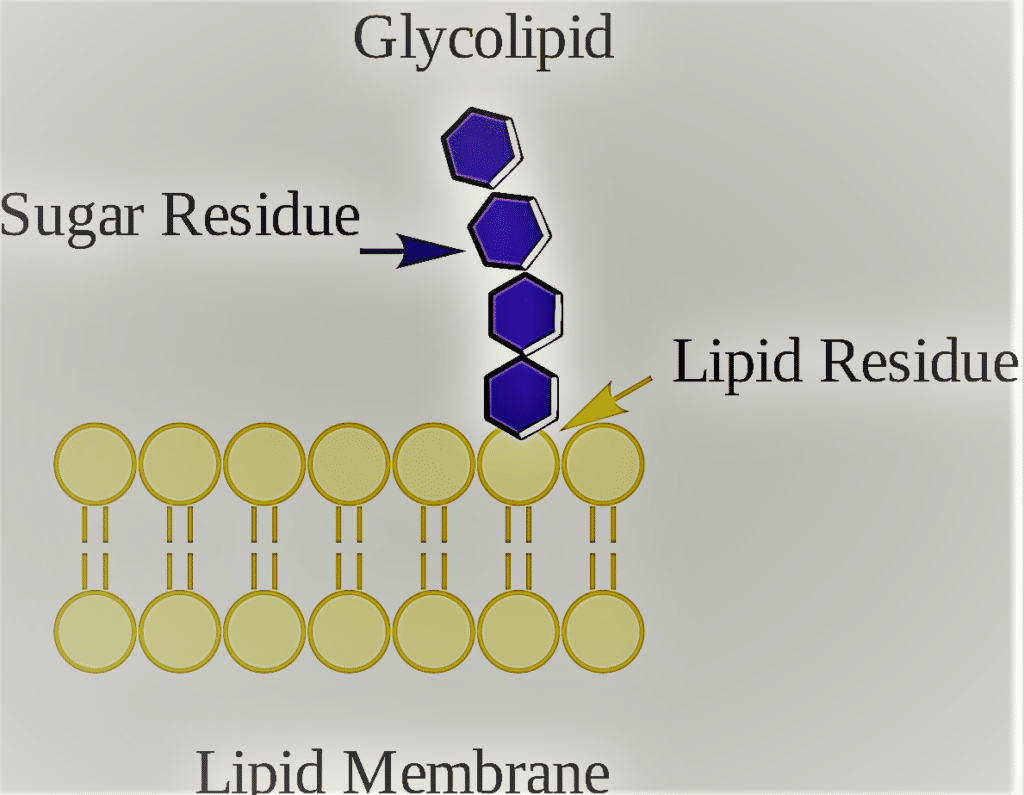
3. Glycolipid
Lipids called glycolipids have a carbohydrate connected via a glycosidic bond. Their job is to keep the cell membrane stability and make it easier for cells to recognize one another, which is important for the immune response and for the connections that allow cells to join together to form tissues.
The outside leaflet of cellular membranes typically contains glycolipids, which serve as receptors, protein anchors, and signal transduction regulators in addition to their structural function in maintaining membrane stability.
Function of glycoconjugates
- Glycoproteins support a variety of physiological processes, including immunity.
- Glycoproteins are necessary for reproduction because they make it easier for sperm cells to connect to the surface of the egg. The mucus contains proteins known as mucins. The chemicals in the digestive, reproductive, urinary, and respiratory tracts shield sensitive epithelium surfaces.
- Glycoproteins play a role in the construction, development, immune system, hormone production, and defense of cells and organisms.
- They identify and bind to lectins, which are carbohydrate receptors on nearby cells. This results in cell-cell attachment and intracellular reactions in the interacting cells.
MCQs/FAQs
What are glycoconjugates?
Covalently joining polysaccharides antigen to protein scaffolds results in glycoconjugates, which are used to induce a sustained immunological response in the body.
Example of glycoconjugates
glycolipid, glycoprotein, proteoglycan, etc