Table of Contents
TogglePauson-Khand Reaction, examples, mechanisms, and applications in organic chemistry have been discussed here:
Pauson-Khand Reaction
The ring formation reaction involves [2+2+1] cycloaddition between an alkyne, an alkene, and carbon monoxide to form α, β-cyclopentenone known as Pauson-Khand reaction. In other words, it is one step-synthesis α, β-unsaturated cyclopentenone from the [2+2+1] cycloaddition of alkyne, alkene, and carbon monoxide, via an intermediate of alkynedicobalt hexacarbonyl complex. Pauson-Khand reaction is also known as Pauson-Khand cyclization, Pauson-Khand cycloaddition, or Pauson-Khand annulation.
Several Pauson-Khand reactions are intended as intramolecular reactions in order to attain stereochemical control. In the case of intermolecular Pauson-Khand reaction, due to steric hindrance, an alkene is positioned next to the less bulky acetylenic substituent during coordination, and a subsequent C-C bond forms between an alkene carbon and the closer acetylenic carbon, leading to the formation of the larger group on alkyne being found at the α-position of the resulting cyclopentenone.
The Pauson-Khand reaction has also been performed under a variety of conditions, including the use of chiral ligands, aldehydes as a carbon monoxide source, solid-supported cobalt catalysts to improve purification, colloidal cobalt nanoparticles, and so on.
The pauson-Khand reaction can be represented as:
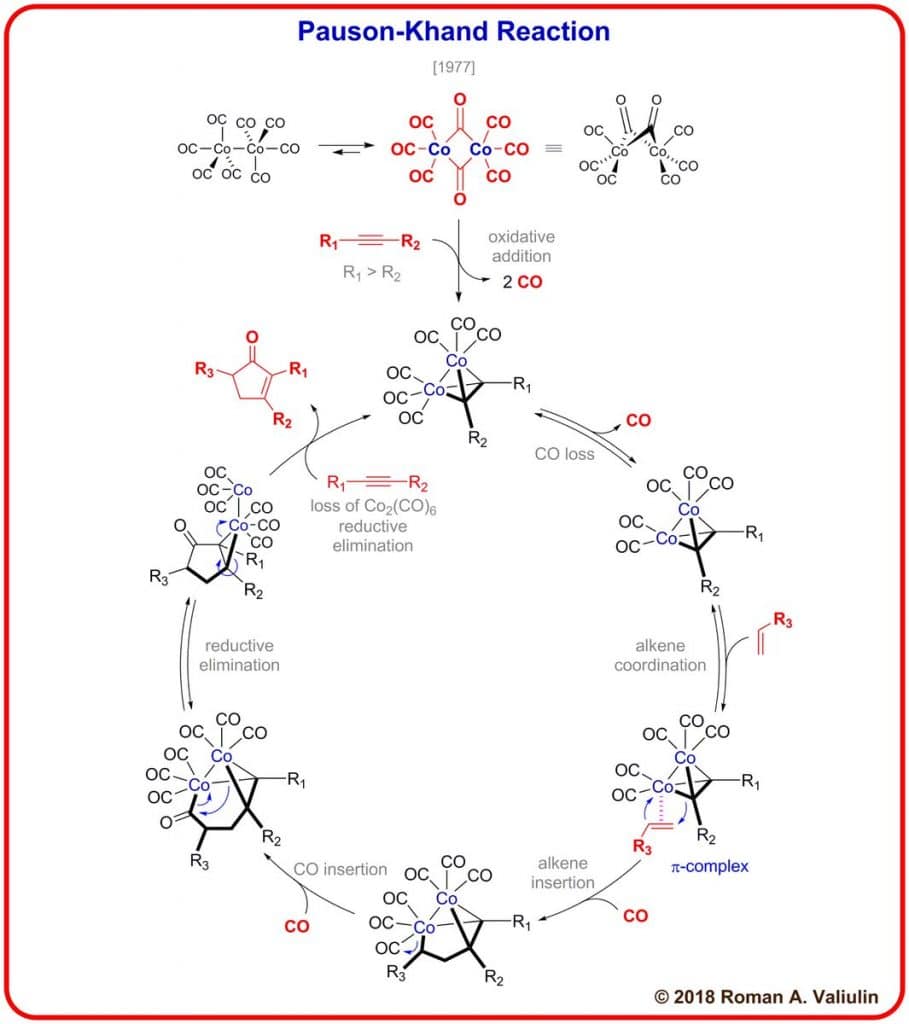
Pauson-Khand Reaction Mechanism
The mechanism of the Pauson-Khand reaction can be carried out in the following way:
Applications of Pauson-Khand reaction
The Pauson–Khand reaction is commonly used to prepare cyclopentenones and their derivatives. This reaction also aids in the overall synthesis of a number of topologically complicated natural products with a variety of structural characteristics.
Pauson-Khand Reaction Video
FAQs/MCQs
What is Pauson-Khand reaction?
Pauson-Khand reaction is [2+2+1] cycloaddition between an alkyne, an alkene, and carbon monoxide to form α, β-cyclopentenone.
Uses of Pauson-Khand reaction
Pauson-Khand reaction is used to prepare cyclopentenone.
References
- March, J., Advanced Organic Chemistry, Wiley Eastern Limited, 1986.
- Skyes, P., A Guide Book to Mechanism in Organic Chemistry, Second edition, Orient Longman Ltd., 1988.
- Ahmar, M.; Knoke, M.; de Meijere, A. and Cazes, B., Synthesis, 2007, 442.
- Perez-Serrano, L.; Blanco-Urgoiti, J.; Casarrubios, L.; Dom ´ ´ınguez, G. and Perez-Castells, J., ´J. Org. Chem., 2000, 65, 3513
- Jeong, N.; Hwang, S. H.; Lee, Y. W. and Lim, J. S., J. Am. Chem. Soc., 1997, 119, 10549.






