Table of Contents
ToggleMichael reaction, examples, mechanism, and applications have been discussed here. This reaction was first of all reported by Michael in 1887.
Michael addition reaction
Michael reaction or Michael addition is the 1,4- addition of a nucleophile to an electron-deficient alkene, alkyne, or α-,β-unsaturated system in presence of a base. This is also known as conjugated addition.
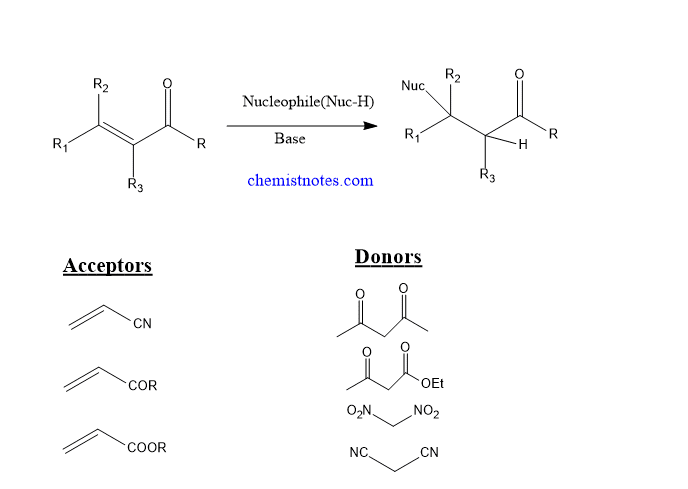
- The nucleophiles are called donors.Malonates, nitroalkanes, cyanoacetic esters, acetoacetic esters, and various β-ketoesters are all donors.
- Electron deficient alkenes or alkynes or α-,β-unsaturated system act as acceptor.
- Initially, unsaturated carbonyl compounds were utilized, but this was later expanded to include other forms of electron deficient alkenes or alkynes.
- In general, Michael addition reactions take place in neutral medium at room temperature without the use of a catalyst.
The addition of a CH-acidic reagent to the carbon-carbon double bond of an α-,β -unsaturated carbonyl compound is the overall procedure.
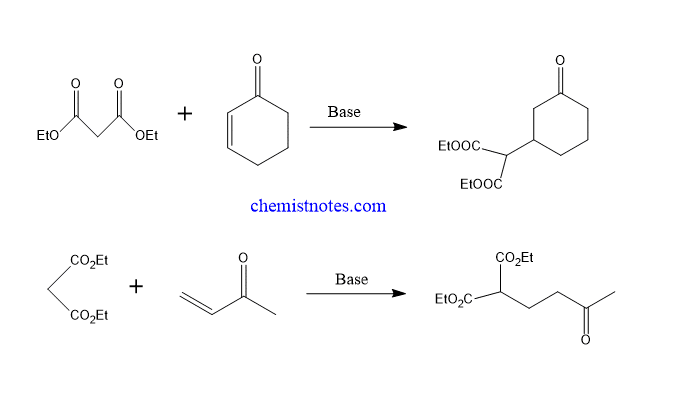
Michael addition reaction examples
Let’s see some examples of this reaction.
Michael reaction mechanism
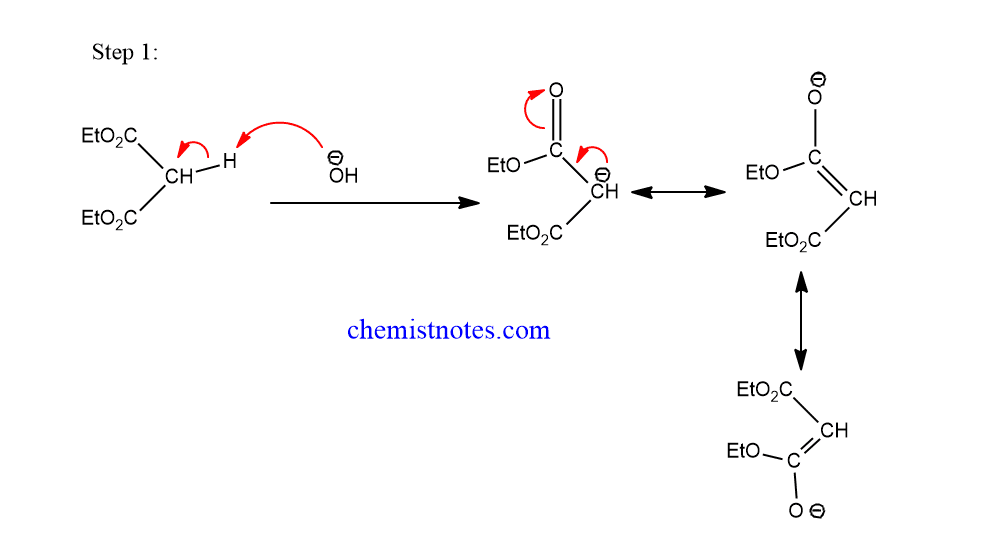
Michael reaction mechanism completes in the following steps:
Step 1: The base abstracts alpha-acidic hydrogen from the nucleophile to form carbanion which is resonance stabilized.
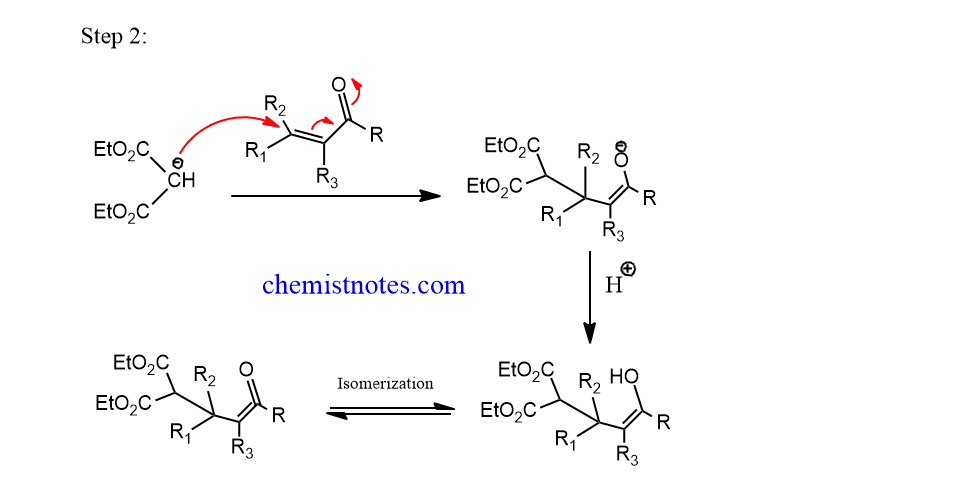
Step 2: The carbanion attacks the electron-deficient compounds.
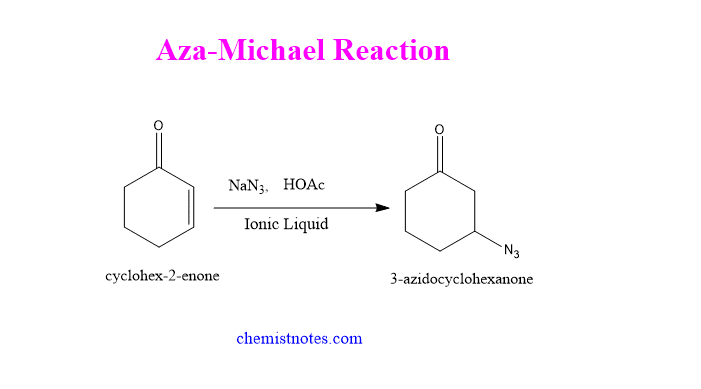
Aza michael reaction
When α-,β -unsaturated carbonyl compound reacts with sodium azide in presence of ionic liquids and Bronsted base as a catalyst, gives an addition product. Such a reaction is known as the Aza-Michael reaction. Let’s see an example:
Application of michael addition reaction
The Michael addition reaction is a very useful organic reaction and it is used in the synthesis of natural products and drugs in organic chemistry.
References:
- Laue T.,Plagens A.,Named Organic Reactions, Second edition, John Wiley & Sons, Ltd, 2005
- Bradford P. Mundy, Michael G. Ellerd, Frank G. Favaloro, Jr., Name Reaction and Reagents in Organic Synthesis, John Wiley & Sons, 2005
- Wang, Z., Comprehensive Organic Name Reactions and Reagents, John Wiley & Sons, Inc.,2010
- J.J. Li, Name Reactions, 4th ed.,© Springer-Verlag Berlin Heidelberg 2009
Please comment below if you find any mistakes or if you have any problems related to this topic. Thank you and happy learning.






