Table of Contents
ToggleChemical bonding is a force of attraction that binds the atoms of the elements together. In other words, the force which binds atoms of elements together in a compound is called a chemical bond.
Cause of chemical bonding(Formation of chemical bonding)
The atoms of the elements other than noble gases tend to remain in a combined state for their stabilities. Such atoms tend to remain in bonded form. They are more stable in bonded form than in uncombined state because the energy of the system is decreased in combined form. Atoms of elements tend to combine with other atoms of elements by losing or gaining or sharing the available valence electrons, which results in the formation of chemical bonding. The tendency of such atoms to gain stability is the reason for the chemical bonding.
Types of Chemical bonding
There are mainly 3 types of chemical bonding
- Ionic bond/Electrovalent bond
- Covalent bond
- Coordinate covalent bond/Coordinate bond
- Ionic bond: The bond formed between two or more than two atoms by the transfer of one or more electrons among atoms is called an ionic bond. Example: Bonding in NaCl, Bonding in Na2O etc.
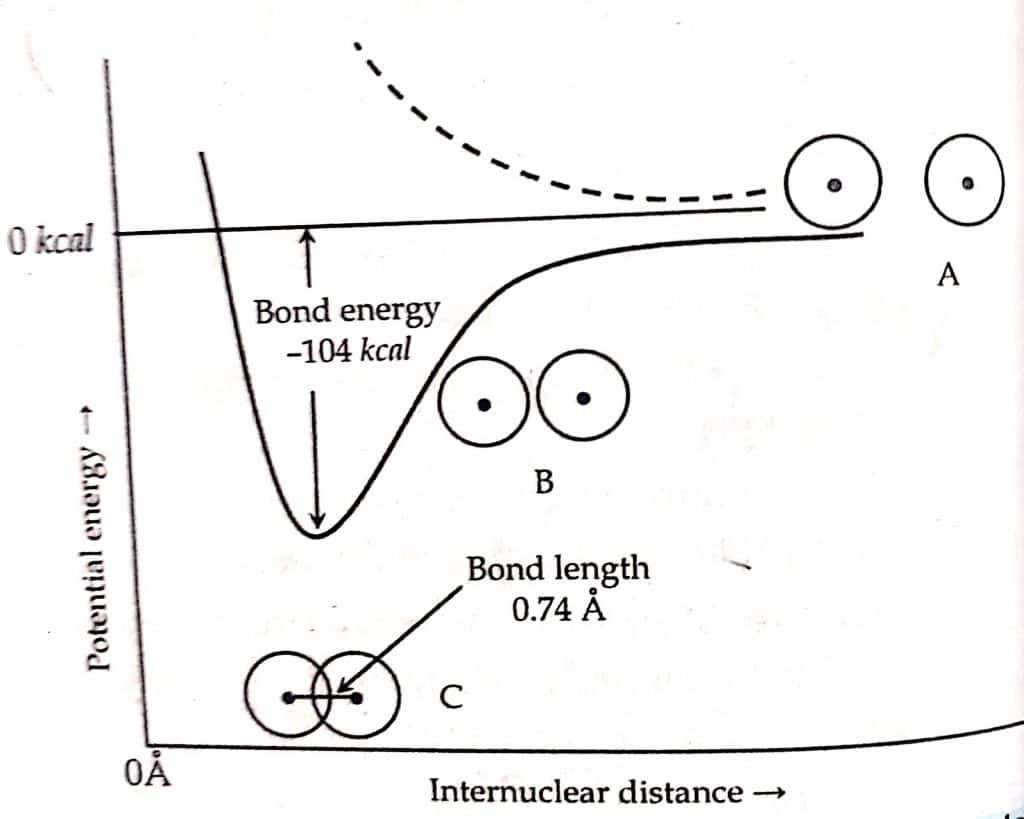
- Covalent bond: A bond formed by the mutual sharing of electrons between the combining atoms of same or different element is called a covalent bond. Example: Bonding in H2, Bonding in HCl etc.
- Coordinate covalent bond: The bond formed by the sharing of electrons between combining atoms in which both the shared electrons are contributed by one of the bonded atoms only, is called co-ordinate bond. Example: Bonding in ammonium ion, Bonding in H3O+ ion etc.
Electronic theory of chemical bond(valency)
G.N Lewis and W. kossel in 1916, published their views independently about the formation of chemical bonds, which are studied together as “Electronic Theory of Valency“.
The main postulates of electronic theory of valency are listed below:
- Valency of an element is determined by the number of valence electrons in the atom of the element. This is because, only valence electrons take part in chemical bonding.
- The atoms of elements other than inert gas tend to gain nearest noble gas electronic configuration for their stability.
- The atoms of elements other than noble gases tend to acquire nearest noble gas configuration by sharing of electrons or loss of electron or gain of electrons.
- Number of electrons lost or gained or shared by an atom of an element is called valency of element. This is the electronic concept of the valency.
- By the bond formation among the atoms of the elements, there is decrease in the potential energy of system. This means, there is overall stabilizaion in the bond formation.
Localized and delocalized chemical bonding
- Localized chemical bonding
A localized chemical bonding is a type of chemical bonding in which electrons forming the bond remains between the bonded atoms.
2. Delocalized chemical bonding
A delocalized chemical bonding is a bonding in which electrons forming bonds don’t remain on a single atom but with several atoms. Such electrons are called delocalized electrons.
What is a chemical bond in chemistry?
Chemical bonding is a force of attraction that binds the atoms of the elements together.
What is meant by a chemical bond?
The force which binds atoms of elements together in a compound is called a chemical bond.
What types of chemical bonds are formed by carbon? why?
Carbon atoms form covalent bonds. Carbon atoms have 4 valence electrons and they can’t donate all valence electrons but can share electrons with other atoms hence forming covalent bonds.
Which chemical bond is the weakest?
Ionic bond is the weakest chemical bond.
Strongest chemical bond
Non polar covalent bond is strongest chemical bond.






