Table of Contents
ToggleChemical vapor deposition (CVD), a bottom-up approach, is one of the most frequently used methods for the synthesis of thin films and nanoparticles. It is a process where one or more gaseous adsorption species react or decompose on a hot surface to form stable solid products. Chemical vapor deposition is more difficult than physical vapor deposition but has benefits including the capacity to create pure thin films or nanoparticles, high manufacturing yield, and simplicity in scaling up.
Chemical Vapor Deposition Process
In Chemical vapor deposition (CVD), a thin layer of material is deposited on a substrate by supplying a mixture of reacting gas (volatile compounds such as SiH4, SiCl4, WF6, etc.) and carrier gas (H2, Ar, etc.)
For instance, thin film of Si3N4 can be deposited using SiH4, NH3, and Ar as:
3SiH4 + 4NH3 → Si3N4 + 12H2
Chemical Vapor deposition process involves the following steps:
- Transport of reacting gaseous species to the surface
- Adsorption of the species on the surface
- Heterogenous surface-catalyzed reactions
- Surface diffusion of the species to growth sites
- Nucleation and growth of the film
- Desorption of gaseous reaction products and transportation of reaction products away from the surface
In chemical vapor deposition, the vapor generated from a precursor is thermally broken down or reacts with another precursor vapor or gas that is present inside the evaporation apparatus. Nucleation, condensation, and coagulation of the precursor particles generate solid-state particles.
Particles size obtained from CVD can range from molecular size to hundreds of micrometers. Some of the advantages of gas to particles conversion method include small particle size (a few nm to mm), narrow size distribution, high purity of the particles, etc. However, the formation of hard aggregates due to agglomeration in the gas phase creates difficulty in the synthesis of high-quality bulk materials.
CVD has been frequently used to grow carbon nanotubes, GaN nanowires, and so on.
Types of chemical vapor deposition
Based on the activation sources for the chemical reactions, the deposition process can be categorized
into:
- Thermally activated chemical vapor deposition
- Plasma Enhanced chemical vapor deposition
Thermally Activated CVD
It is a conventional CVD in which gaseous precursors used to deposition are dissociated thermally and deposited on a heated substrate. Because the reaction temperature for this approach must be high, substrates with high melting temperatures that are lower than the reaction temperature cannot be employed. One of the most often utilized heating sources in CVD reactors is the tungsten filament. Using this technique, SiC nanorods, Fe nanoparticles, etc. are being synthesized.
Plasma Enhanced Chemical Vapor Deposition
With a lower deposition temperature than thermal CVD, plasma-enhanced CVD offers a clear advantage. In this process, the plasma produced from several sources, including DC, RF(AC), and microwave, is used to enhance the reaction between the precursors.
In DC plasma, reacting gases are broken down and ionized using a DC electrical discharge to create plasma, while in AC plasma, reacting gases are decomposed and ionized generating a time-varying plasma that is repeatedly initiated and extinguished. Nowadays, RF plasma-enhanced CVD is frequently used to deposit SiC films on silicon substrate using a mixture of SiCl4, CH4, H2, and Ar.
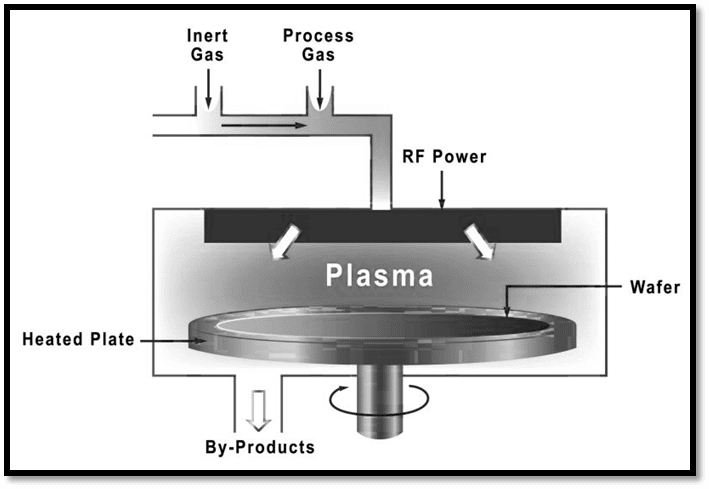
Microwaves are utilized to create plasma in microwave plasma-enhanced CVD (MW-CVD). The main impact of microwaves is to oscillate electrons, and when these electrons collide with gaseous atoms and molecules, they produce significant ionization percentages. Additionally, it provides good substrate-specific selective growth. Arrays of carbon nanotubes that are vertically oriented are typically grown using MW-CVD.
Chemical vapor deposition vs physical vapor deposition
The difference between chemical vapor deposition and physical vapor deposition are:
| Chemical Vapor Deposition | Physical Vapor Deposition |
| Also known as CVD. | Also known as PVD. |
| One or more gaseous adsorption species react or decompose on a hot surface to form stable solid products. | The material is heated above its melting point to generate vapors, which are then deposited on the target surface. |
| Gaseous molecules react with the substrate. | On the substrate, atoms are moving and deposit. |
| Deposition is carried out either by thermally activated CVD or plasma-enhanced CVD. | Vapors are generated using sputtering, evaporation, electron beam, wire explosion, and so on. |
| CVD processes are restricted to two active gases and do not result in coating on the substrate through a high-temperature reaction or gas phase. | Physical vapor deposition methods, which include warm or hot in vacuum methods, are extremely varied. |
Advantages of chemical vapor deposition
- It demonstrates a number of distinct advantages, including the ability to generate highly pure and dense films or small particles at comparatively high deposition rates and the capacity to evenly coat components with complex shapes due to its non-line-of-sight nature.
- A variety of metallic, ceramic, and semiconducting thin films are being deposited by CVD.
Disadvantages of chemical vapor deposition
- Due to variations in vapor pressure, nucleation, and growth rates during the gas-to-particle conversion, which all result in the heterogeneous composition of particles, it is challenging to synthesize multi-component materials by CVD.
- Absence of extremely volatile, nontoxic, and nonpyrophoric precursors in thermally activated CVD.
FAQs
What is chemical vapor deposition
Chemical vapor deposition is a bottom-up based synthetic approach for the preparation of thin films and nanoparticles
.
Chemical vapor deposition equipment
Typical chemical vapor deposition equipment includes a gas delivery system, reaction chamber or reactor, loading/unloading system, energy source, vacuum system, process automatic control system, and exhaust gas treatment system.
Chemical vapor deposition video
References
- Piszczek, P., & Radtke, A. (2018). Silver Nanoparticles Fabricated Using Chemical Vapor Deposition and Atomic Layer Deposition Techniques: Properties, Applications and Perspectives: Review. Noble and Precious Metals – Properties, Nanoscale Effects and Applications. doi: 10.5772/intechopen.71571
- Dobkin and Zuraw (2003). Principles of Chemical vapor Deposition. Kluwer. ISBN 1-4020-1248-9.