Table of Contents
ToggleThe absolute and relative configurations are the two most important concepts used in stereochemistry In order to understand how molecules are placed together and interact with one another. Stereochemistry is the study of the molecular geometry of atoms in three-dimensional space. The arrangement of the atoms in space of a molecule of the defined constitution without regard to arrangements that differ only as after rotation about one or more single bonds is called configuration (Cross and Klyne, 1976). The determination and specification of configuration are essential for the structure determination of the molecules displaying stereoisomerism including chiral ones.
Absolute configuration refers to the actual arrangement of atoms or groups in space of asymmetric carbon atoms, while relative configuration refers to the correlative configuration that relates the configuration of one compound with another.
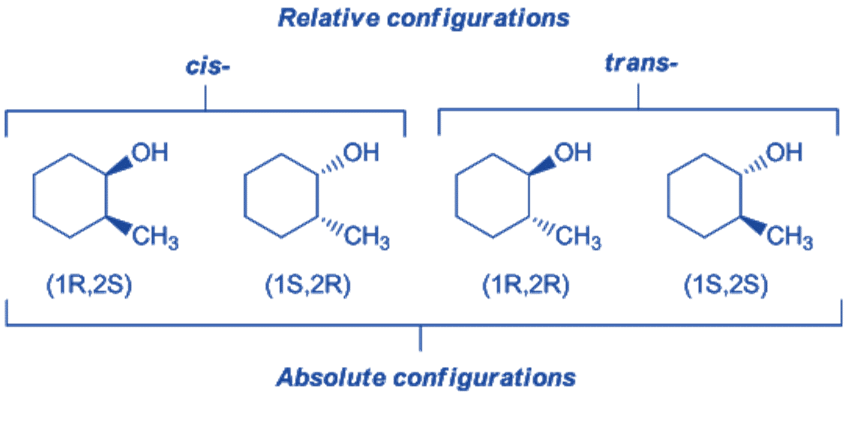
Absolute Configuration
Absolute configuration is the actual/precise configuration of atoms in space. Since each stereoisomer has a unique arrangement of groups on the carbon atoms in space, the stereoisomers exhibit different sequences of the four groups on the asymmetric carbon atom. When the configuration of a given stereoisomer is known, its absolute configuration is said to be known.
Let’s take an asymmetric molecule, Cabcd which has two stereoisomers, and are enantiomers.

In the above structures, one of them has (+) rotation, while another has (-) rotation. It is quite difficult to know which isomer gives (+) rotation and which one gives (-) rotation. Knowing the configuration of the (+) and (-) compounds involves also knowing the absolute configuration of that compound.
Absolute configuration is indicated by R and S configuration (Cahn-Ingold-Prelog rules).
R and S Configuration
R.S. Chan, C.K. Ingold, and V. Prelog have introduced a new convention to give the configuration of an asymmetric carbon atom. This is known as R and S configuration. If the priority order is given to maintain the order of groups around the asymmetric carbon atoms, then the rule is called the sequence rule.
The basic rules for R and S configuration are:
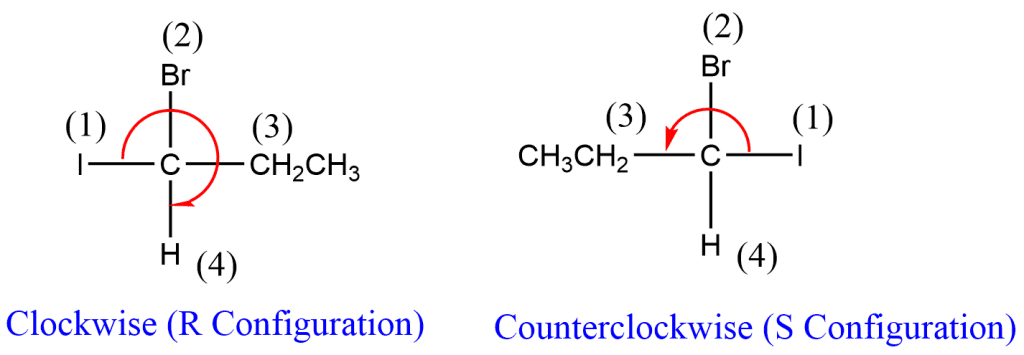
- The optically active molecule should be shown in three dimensions. The group that comes last on the priority list should be pointed away from the spectators. Therefore, as indicated in the image, when looking down at the molecule, the group with the lowest priority will not be visible.

2. Observe whether the path is clockwise (rectus or R) or counter-clockwise (sinister or S). The compound is considered to have R configuration if the path taken travels clockwise from the highest priority group to the lowest one. In a similar manner, if the priority order has moved counterclockwise or from lowest to highest, it is known as sinister, and the configuration is referred to as an S configuration. The R and S system of configuration can also be used to provide the nomenclature of the compound. In Fischer’s projection formula, the group with the lowest priority should be at the bottom.
A priority order or set of sequence rules has been established as follows:
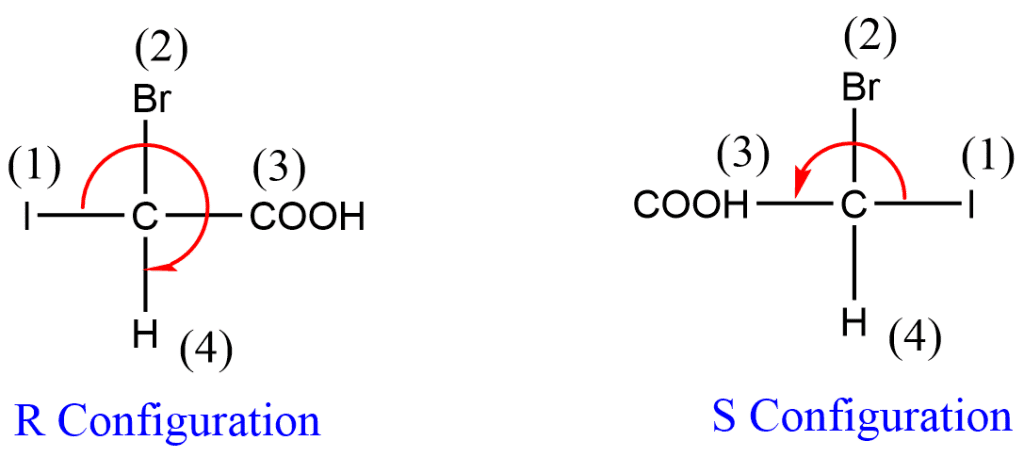
(i) Susbstituents are listed in order of decreasing the atomic number of the atoms which are directly joined to an asymmetric carbon atom.
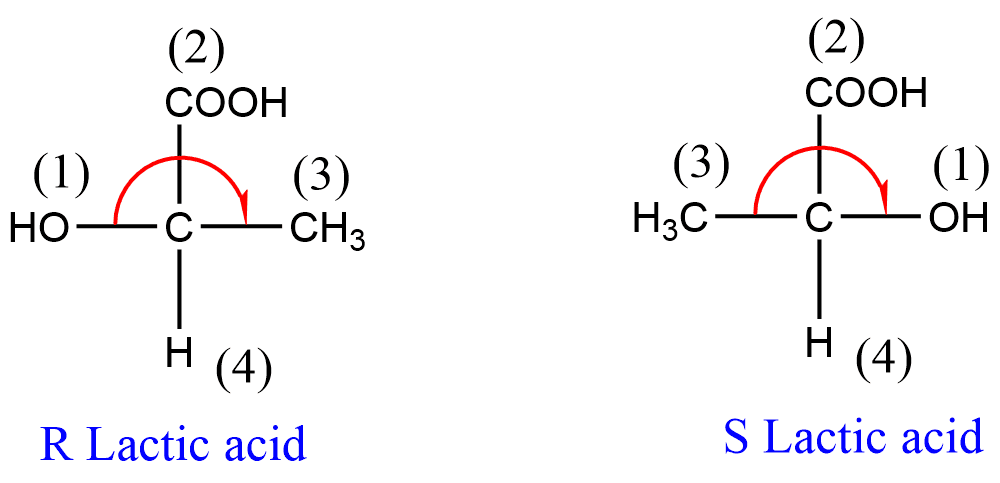
(ii) If there are two or more identical atoms directly linked to the asymmetric carbon, the priority order is determined by the atomic number of the second atom. For example, in lactic acid, there exist COOH and CH3 groups directly attached to an asymmetric carbon atom. Since the atomic number of the first element of both these groups is the same (i.e. 6 for carbon), the priority order of these groups will be determined by the second atoms i.e. O-atom of -COOH group and H-atom of the CH3 group. Because of the fact that Oxygen atom of COOH group has higher atomic number than the hydrogen atom of CH3 group, -COOH takes priority over -CH3.
(iii) If the first and second sets of atoms are identical in two or more groups, the group with the highest atomic number of substituents comes first. For example: -CH(OCH3)2 > -CH2OCH3; -CHCl2 > CH2Cl, and so on.
(iv) The atoms with multiple bonds are counted twice or three times when they are joined to the asymmetric carbon atom by double or triple bonds. This is the reason that the oxygen atom in C=O group is counted twice. Due to the groups having multiple bonds, the priority order of some groups are like: -CH=O>-CH2OH.
Several methods, including X-ray crystallography, nuclear magnetic resonance spectroscopy, and electron diffraction, can be used to determine the absolute configuration of a molecule.
Relative configuration
Relative configuration depicts the arrangement of atoms or groups of atoms attached to the asymmetric carbon atom in relation to the arrangement of atoms or groups of atoms attached to the asymmetric carbon atom of another molecule. In the relative configuration, the relation of the configuration of one molecule is established with another molecule. It is independent of rotating the plane polarized light as well as R and S notations.
If one molecule is converted into another molecule without breaking the bond at the chiral center, then the molecules are said to have the same relative configuration. For example, conversion of D(+)-Glyceraldehyde into D(-)-Glyceric acid.

Here, the glyceric acid is formed from D(+)-Glyceraldehyde without breaking the bond at the chiral center. Hence it establishes the relation of configuration with D(+)-Glyceraldehyde. Therefore, the glyceric acid thus formed has D configuration i.e. the relative configuration of a compound with D(+)Glyceraldehyde also results D configuration.
Let us consider the conversion of D(+)-Glyceraldehyde to (+)- and (-)-malic acid separately without breaking the bond at a chiral center. It shows that D(+)-Glyceraldehyde has established the relative configuration with both (+)-malic acid as well as (-)-malic acid.
From the above illustration, it becomes clear that both malic acids can be prepared from D(+)-Glyceraldehyde. Here, D(+)-Glyceraldehyde has established relative configuration with both compounds. As a result, (+)- and (-)-malic acid has the same ‘D’ configuration as D(+)-Glyceraldehyde from which it is prepared without breaking the bond at the chiral center. In such cases, the reactant and product have the same configuration. Thus, the relative configuration is based on the relationship of one molecule with another. However, the Specifications of R and S notations in these molecules are required if we want to observe the absolute configuration of the molecule.
Moreover, all absolute configurations are relative configurations but not all relative configurations are absolute configurations.
Nuclear magnetic resonance spectroscopy is one method for determining the relative configuration.
Difference between relative and absolute configuration
| Absolute Configuration | Relative Configuration |
| Absolute configuration is the actual arrangement of atoms or groups of atoms in space. | Relative configuration is the arrangement of atoms or groups of atoms relative to the arrangement of another compound. |
| Does not depend on the position of other substituents. | Since one molecule establishes relation with another molecule, relative configuration depends on the position of other substituents. |
| R and S notation signifies absolute configuration. | R and S configuration is not concerned in relative configuration. |
Absolute and Relative Configuration Video
References
- E. L. Eliel, S. H. Wilen and L. N. Mander, Stereochemistry of Organic Compound, John Wiley & Sons, Inc., 1994.
- K. Mislow, Introduction to Stereochemistry, W.A. Benjamin Inc, 1966.
- K. R. Palak, Stereochemistry, Pairavi Prakashan, Kathmandu, Nepal, 2017.






