Table of Contents
ToggleWhat is active site of enzyme? You may have heard about these sites in several research papers. The active site is a specific area of the enzyme that is responsible for substrate binding and catalysis. Enzymes are large in size compared to substrates which are relatively smaller. Let’s discuss the active sites and their features in a brief.
what is active site of enzyme?
Active site of an enzyme
An enzyme’s active site or active center is a tiny region of the enzyme where the substrate binds and participates in catalysis. An enzyme’s active site is the area that binds the substrate and converts it to the product. It is a three-dimensional entity created from amino acid residues that might lie far apart in the linear polypeptide chain and is usually a very tiny part of the entire enzyme molecule.
Moreover, the active site is commonly a cleft on the surface of the enzyme that produces a largely non-polar environment, which improves substrate binding. Multiple weak forces bind the substrate in the active site, including electrostatic contact, hydrogen bonds, van der wall bonds, and hydrophobic interaction.
Characteristics of active site of enzyme
The characteristics or features of the active sites of the enzyme are described below:
- The presence of an active site is related to the protein’s tertiary structure, which results in a three-dimensional native conformation.
- An active site is defined as a cleft or pocket that occupies a small portion of a large enzyme molecule.
- The active site is composed of catalytic residues, which are located far apart in the linear sequences of amino acids.
- The structure and geometry of the active sites are not rigid. It is rather adaptable in order to facilitate specific substrate binding.
- In general, active sites have a substrate-binding site and a catalytic site. The catalytic site is responsible for catalyzing the specified reaction.
- The substrate forms weak noncovalent linkages with the active site, such as van der Waal forces, hydrogen bonds, electrostatic force, etc.
- Because of the presence of active sites, enzymes are specialized in their function.
- Serine, aspartate, cysteine, lysine, histidine, arginine, glutamate, tyrosine, and other amino acids are typically present at active sites. Serine is the most common amino acid among these.
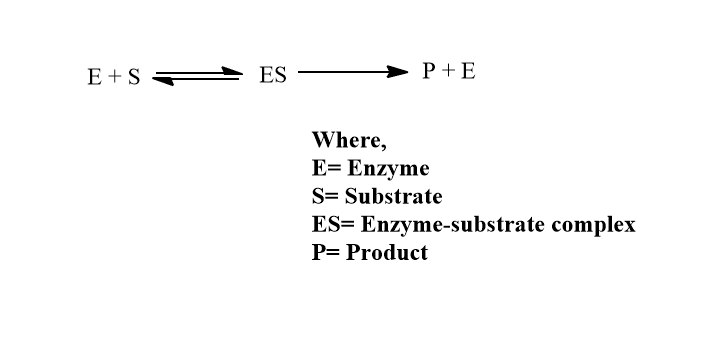
- The substrate (S) interacts with the enzyme (E ) at the active site, resulting in the formation of the enzyme-substrate complex (ES). After the catalysis, the product P is released, and the enzyme is ready for reuse.