Table of Contents
ToggleMcFarland Turbidity is generally used to measure turbidity in the case of antimicrobial tests. McFarland is the composition of barium chloride and sulphuric acid. It is generally employed for the balance of approximately the bacterial colony or bacterial suspension in the plates.
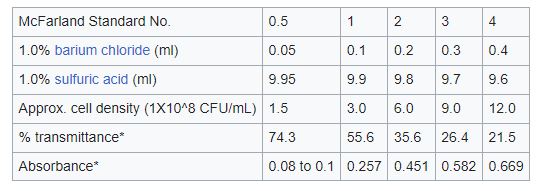
Mcfarland standard
To standardize microbial testing, McFarland Turbidity standards are used as a guide to altering the turbidity of bacterial suspensions so that the number of bacteria will be within a specific range. Antibiotic susceptibility testing by determination of the lowest inhibitory concentration, which is frequently used in medical microbiology and research, is an illustration of such testing. For any given antimicrobial agent, an incorrect outcome (falsely resistant or falsely susceptible) could happen if a suspension is employed that is too heavy or too dilute.

History of McFarland turbidity
The McFarland turbidity test, commonly referred to as the McFarland scale or McFarland standard, is a method used in microbiology to gauge the density of bacterial suspensions. Dr. J.H. McFarland, an American microbiologist, discovered it in 1907.
In the McFarland turbidity test, a bacterial suspension’s turbidity or cloudiness is contrasted with several reference barium sulfate solutions. The known turbidities of the barium sulfate solutions, which range from 0.5 to 8.0 McFarland units, are utilized as a guide to calculate the density of the bacterial suspension.
Principle of the McFarland test
The McFarland test is based on the idea that a bacterial suspension’s turbidity is directly inversely related to the number of bacteria in the suspension. During the test, a standardized turbidity standard is used, which is a barium sulfate suspension in sulfuric acid solution with a predetermined optical density at a specific wavelength of light.
Preparation of McFarland standard
Protocol:
- Barium sulfate powder
- 1.175% hydrochloric acid (HCl) solution
- Distilled water
- Graduated cylinder or pipette
- Sterile test tubes or vials
- Initially, distilled water is used to dissolve the barium sulfate powder to prepare a saturated solution.
2. To make a 0.5 McFarland standard, the saturated solution is diluted with distilled water. To do this, combine one milliliter of the saturated solution with 99 milliliters of distilled water. Mix thoroughly.
3. To make a 1.0 McFarland standard, add 1 ml of the 0.5 McFarland standard to 99 ml of distilled water, stirring well after each addition.
4. To create standards with larger concentrations, such as 1.5, 2.0, 2.5, 3.0, 4.0, 5.0, 6.0, 7.0, and 8.0 McFarland, the process is repeated.
5. The proper McFarland unit concentration will then be written on each standard, and it will be kept in a cold place.
How to perform the McFarland test?
A small amount of the bacterial culture is initially put into a tube of sterile saline solution to conduct the McFarland turbidity test. The tube is then contrasted with a group of standardized tubes that hold the turbidity standard. To dilute the bacterial culture to a particular cell density, the tubes are brought up to a light source, and the tube most closely fits the turbidity standard is chosen.
Application of Mcfarland test?
- Testing for antibiotic susceptibility:
- The McFarland turbidity test is frequently used to set the bacterial inoculum for testing for antibiotic susceptibility standards. For accurate and repeatable test findings, the standardized inoculum ensures that the bacterial culture has a constant cell density.
- Microbiology laboratories also employ the McFarland turbidity test for quality assurance.
- The test aids in ensuring that test findings are accurate and dependable by making sure that the bacterial cultures used in testing have a consistent cell density.
- Blood culture testing:
- The bacterial inoculum for blood culture testing can be standardized using the McFarland turbidity test. This lessens the possibility of false negative results and increases the test’s sensitivity.
- Diagnostic testing:
- Particularly for bacterial infections, the McFarland turbidity test is employed in diagnostic testing. The test aids in ensuring that the culture is optimal for the identification of the infectious agent by standardizing the bacterial inoculum.